- Health
Claim that the HPV vaccine increases risk of cervical cancer misleads by cherry-picking data from FDA document
Key takeaway
According to the World Health Organization, cervical cancer is the fourth most common cancer in women worldwide. In 2022, there were approximately 660,000 new cases of cervical cancer and around 350,000 deaths from cervical cancer. Infection by the sexually-transmitted human papillomavirus (HPV) is the main cause of cervical cancer. HPV vaccination can help prevent infection with high-risk HPV types, thereby reducing the risk of cervical cancer.
Reviewed content
Verdict:
Claim:
HPV vaccine increases the risk of developing cervical cancer
Verdict detail
Factually inaccurate: Large-scale published studies have shown that getting the HPV vaccine is associated with a strikingly reduced risk of cervical cancer.
Cherry-picking: The claim is based on one particular table from a document which showed negative vaccine efficacy in people infected with HPV prior to vaccination. However, the document also explained that differences in baseline demographics between the vaccine and placebo groups may have influenced vaccine efficacy measurements. This explanation is typically left out of the claim.
Full Claim
“Did you know if you already had HPV, Gardasil made you more likely to get cervical cancer?”; FDA data shows those who received the HPV vaccine were more likely to get cancer compared to those who weren’t vaccinated
Review
The World Health Organization reports that cervical cancer is the fourth most common cancer in women worldwide. In 2022, there were approximately 660,000 new cases of cervical cancer and around 350,000 deaths from cervical cancer. The main cause of cervical cancer is the sexually-transmitted human papillomavirus (HPV).
HPV vaccines can help prevent infection by high-risk HPV types, thereby reducing the risk of cervical cancer. The vaccine cannot treat existing infection; as such, it’s most effective when given before a person is exposed to HPV, meaning before they begin sexual activity.
However, some opposed to vaccination claim that the HPV vaccine increases a person’s risk of cervical cancer. Among them are nephrologist Suzanne Humphries, as seen in this video from 2016, and obstetrician-gynecologist James Thorp, who tweeted this claim in December 2023.
More recently, in March 2024, critical care physician Pierre Kory—who has spread misinformation about COVID-19 and vaccines in the past—made the same claim in an Instagram post.
Evidence for this claim commonly involves the background document for a May 2006 meeting held by the U.S. Food and Drug Administration (FDA)’s Vaccines and Related Biological Products Advisory Committee (VRBPAC). This FDA committee comprises experts in various fields, such as immunology, virology, and epidemiology, who review the safety and effectiveness of vaccines and other biological products.
The background document shows the VRBPAC’s discussion of results from several clinical trials testing Merck’s HPV vaccine Gardasil. The basis for the claim that Gardasil increases the risk of cervical cancer is in Table 17 of the document, which showed negative vaccine efficacy in people who were already infected with HPV.
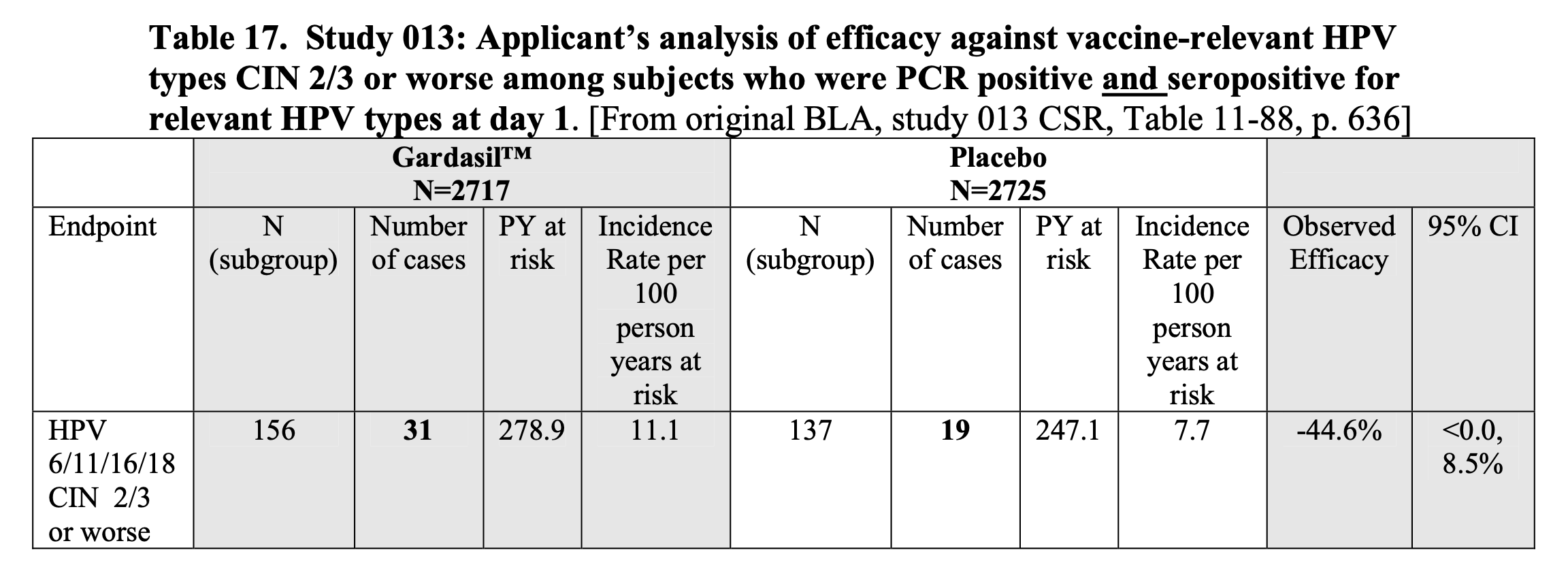
Figure 1 – Screenshot of Table 17 in the 18 May 2006 VRBPAC background document on Gardasil, showing a finding of negative vaccine efficacy in one of the clinical trials.
In brief, vaccinated people in this particular group appeared to have a higher risk of developing precancerous lesions (cervical intraepithelial neoplasia, or CIN for short) compared to unvaccinated people.
However, the claim is an example of cherry-picking information to suit a particular narrative, as a complete reading of the document—rather than the fragment presented in instances of the claim—tells a different story.
The background document showed that Table 17—showing data from the FUTURE I clinical trial—did initially trigger the VRBPAC’s concern that Gardasil could increase the risk of cervical cancer in a subgroup of people who were already infected by HPV before getting vaccinated.
The document also explained that this finding led the FDA’s Center for Biologics Evaluation and Research (CBER) to ask for more information. Specifically it wanted to evaluate whether there were potential imbalances in the baseline demographic characteristics of the Gardasil and placebo groups.
This is important because a finding of negative vaccine efficacy may not be because the vaccine enhances disease risk, but rather due to biases in the study like confounding factors[1]. Also, because the Gardasil and placebo groups were relatively small, potential imbalances in demographics could arise by accident. Additional analyses help to determine if biases could have significantly affected the results.
Figure 2 below shows that CBER examined demographics for the Gardasil and placebo group, including smoking status, history of cervicovaginal infection or sexually transmitted disease, and positive results for a certain type of abnormal cell (high-grade squamous intraepithelial lesion, or HSIL for short) in the cervix prior to vaccination. These are all risk factors for cervical cancer.
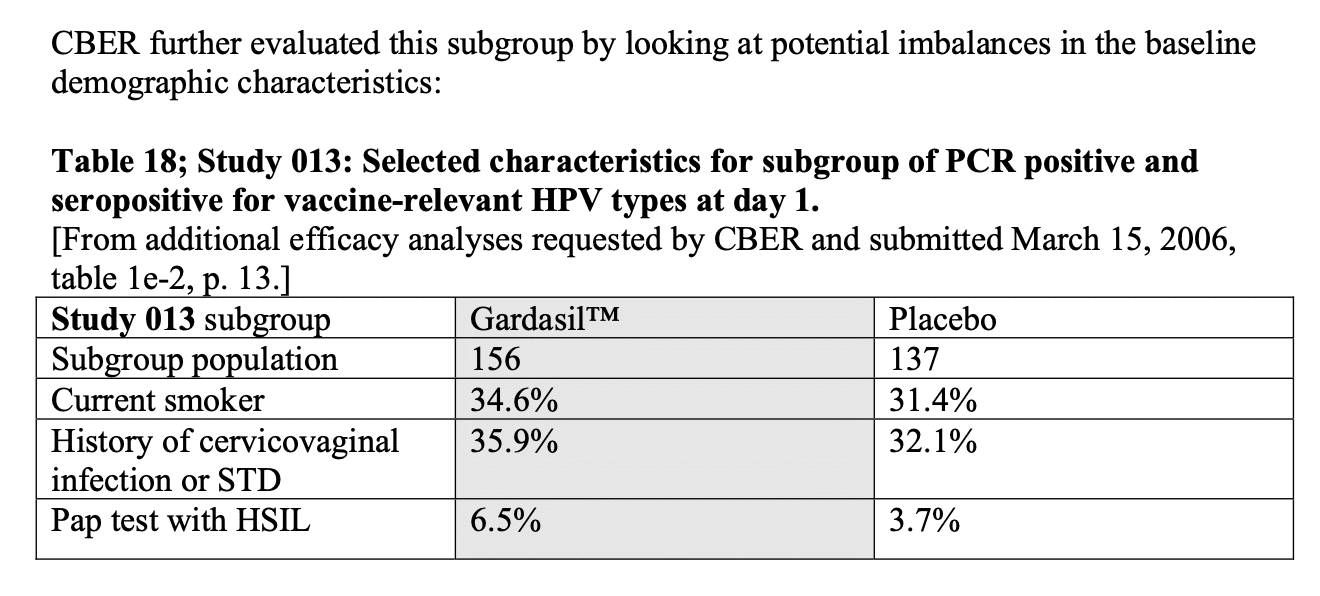
Figure 2 – Screenshot of Table 18 in the 18 May 2006 VRBPAC background document on Gardasil, showing differences in baseline characteristics of the Gardasil and placebo groups.
Based on the demographic data, the VRBPAC concluded that the people with HPV infection prior to vaccination who received Gardasil “might have had enhanced risk factors for development of CIN 2/3 or worse compared to placebo recipients”. CIN 2 and 3 indicate a greater presence of abnormal cells in the cervix and a greater risk of developing cervical cancer.
These risk factors could explain why the Gardasil group appeared to have a greater risk of developing CIN compared to the placebo group.
The VRBPAC also considered data from a later clinical trial (FUTURE II), which is shown in Table 20 of the background document. As Figure 3 below shows, data from this clinical trial didn’t show negative vaccine efficacy in people who were already infected with HPV, unlike data from FUTURE I. The VRBPAC also added that “the baseline demographic data appeared to be more balanced in this subgroup” in FUTURE II.
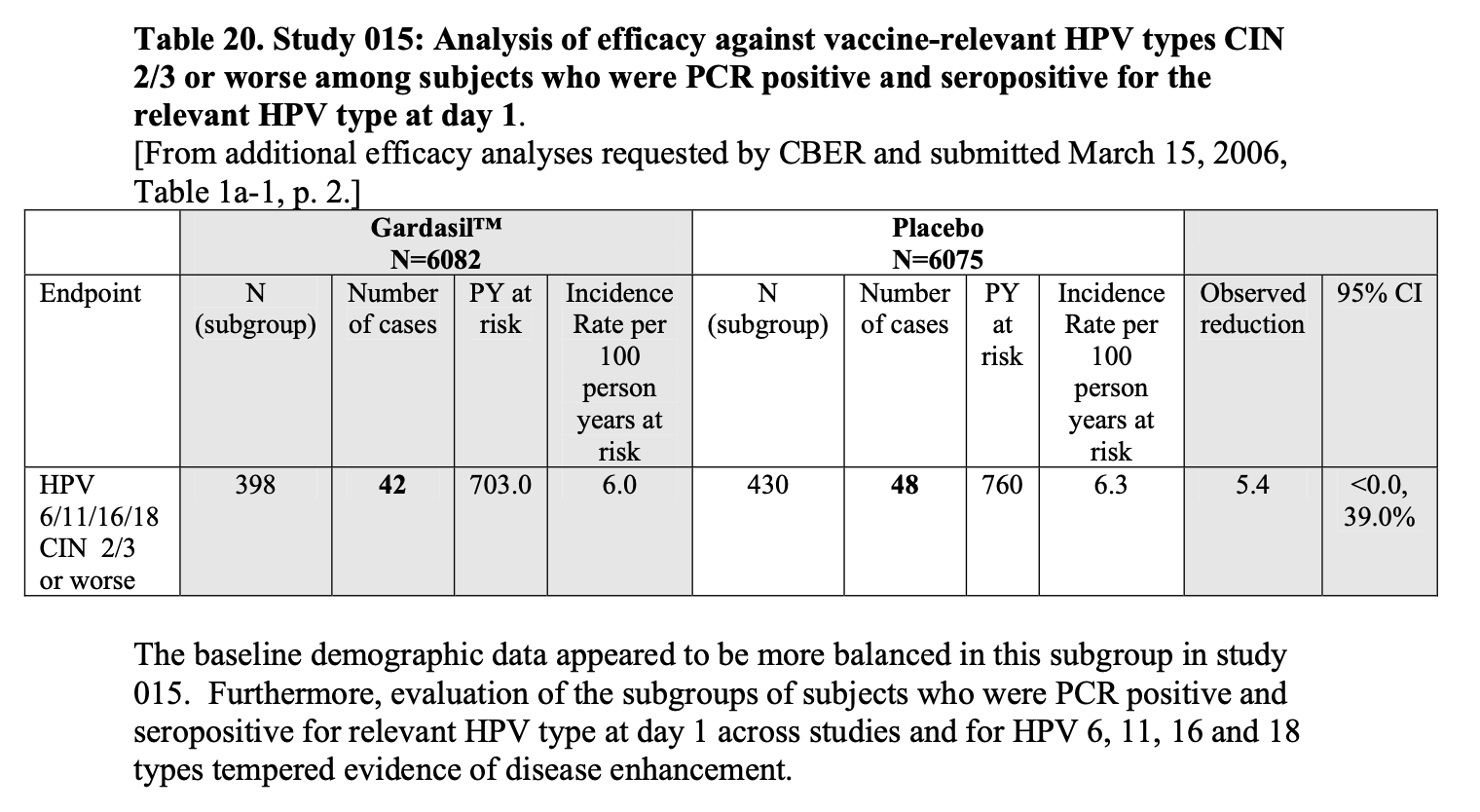
Figure 3 – Screenshot of Table 20 in the 18 May 2006 VRBPAC background document on Gardasil, showing vaccine efficacy in people infected with HPV prior to vaccination. These results were from a later study (FUTURE II).
From this, we can see how posts claiming that the document showed the HPV vaccine increased cancer risk left out important findings shared by Tables 18 and 20, lending Table 17 exaggerated importance. These posts also didn’t show the full context around the VRBPAC discussion, which would have led to a different conclusion than the one shown in the posts.
Real-world data shows the benefits of HPV vaccine
Moreover, the 2006 VRBPAC meeting isn’t the sole source of information on how the HPV vaccine influences cervical cancer risk. Large published studies have since shown that, in the real world, getting the HPV vaccine is associated with a lower risk of developing cervical cancer[2,3].
For example, a study in Sweden, which examined 1.7 million women over 11 years, found that cervical cancer incidence was reduced by approximately 53% in women who received their first dose between 17 and 30 years old. This reduction in cancer incidence became even more striking in women who received their first dose before the age of 17: an almost 90% reduction in cervical cancer incidence in vaccinated women compared to unvaccinated women[2].
A study in Scotland examined the medical records of approximately 430,000 women born between 1988 and 1996[3]. Among them, 40,000 were vaccinated between 12 and 13 years of age, while 124,000 received the vaccine at 14 or older. The rest were unvaccinated.
The researchers observed no cervical cancer cases in women who were vaccinated before they turned 14. Moreover, this was the case even for women who received fewer than three doses, which was the recommended number of doses to complete HPV vaccination in Scotland at the time period studied.
While the researchers did observe cervical cancer cases in women who were vaccinated at 14 years of age or older, the incidence in this group (3.2 cases in 100,000 people) was less than half of that observed in unvaccinated women (8.4 cases in 100,000).
Conclusion
In summary, data from an FDA document doesn’t demonstrate that the HPV vaccine increases the risk of cervical cancer, contrary to anti-vaccine claims on social media. These claims exaggerate the importance of a particular analysis and ignore information in the same document explaining the drawbacks of that analysis.
Large published studies don’t support the claim that the HPV vaccine increases the risk of cervical cancer. On the contrary, they show that getting the HPV vaccine is associated with a lower risk of developing cervical cancer, and this effect is enhanced when people get vaccinated before beginning sexual activity.
REFERENCES
- 1 – Bodner et al. (2023) Observed negative vaccine effectiveness could be the canary in the coal mine for biases in observational COVID-19 studies. International Journal of Infectious Diseases.
- 2 – Lei et al. (2020) HPV Vaccination and the Risk of Invasive Cervical Cancer. New England Journal of Medicine.
- 3 – Palmer et al. (2024) Invasive cervical cancer incidence following bivalent human papillomavirus vaccination: a population-based observational study of age at immunization, dose, and deprivation. Journal of the National Cancer Institute.